|
9/19/2023 0 Comments Hcn molecular geometry 
Hydrogen has one, carbon contains four, and nitrogen has five electrons around its atom. Place the Valence Electrons around the Atoms As carbon is least electronegative than nitrogen, so, the carbon atom wins the central place here. You can select the center atom from either carbon or nitrogen. It’s because the hydrogen atom has only one electron, it cannot form a bond with two atoms (carbon and nitrogen). Here, hydrogen cannot serve as the central atom. The electronegativity of hydrogen, carbon, and nitrogen is 2.2, 2.55, and 3.04, respectively. Hence, the valence electrons in HCN = 10.Īccording to the rule, the atom with the lowest electronegativity value would occupy a central place. To calculate the total valence electrons, add the valence electrons from all three atoms the total number of valence electrons present is 1+4+5=10. 
Therefore, nitrogen has five valence electrons. of atom (1) = 4Īs nitrogen is a VA element in the modern periodic table, Therefore, the valence electrons in the outermost shell of carbon are four. of atom (1) = 1Īs carbon is an IVA element of the periodic table, Hence, it has only one electron in its valence shell. Hydrogen belongs to the IA group of the modern periodic table. 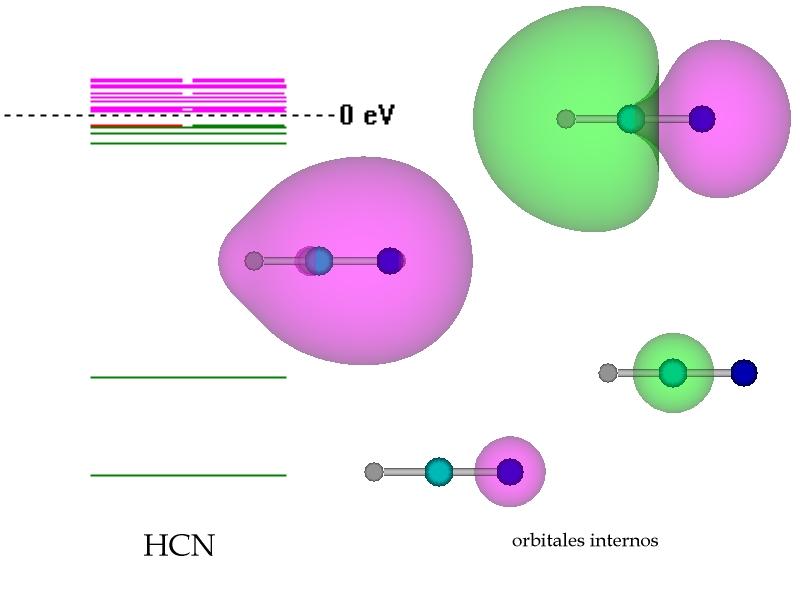
Now let us calculate the Valence Shell Electron of hydrogen cyanide. NOTE: To calculate the total outermost electrons of a particular element in molecule, multiply the number of atoms of particular element by its valence shell electrons. Carbon belongs to group IVA element with four electrons in its outermost shell, while Nitrogen is in-group VA of the modern periodic table with five electrons in its outermost shell. Hydrogen lies in the IA group of the periodic table with one valence electron. While drawing the HCN lewis structure we should know that hydrogen cyanide is made of three elements: hydrogen, carbon, and nitrogen. To Determine the Total Number of Valence Electrons A dot represents a lone pair of electrons in a Lewis structure, whereas a straight line indicates a bond pair of electrons. The Lewis structure merely takes into account the electrons present in the valence shell, neglecting the inner shell’s electrons. Calculate the total number of electrons in the valence shells.ĭraw the HCN Lewis Structure in 3 Easy Steps.We’ll look at each stage of drawing the HCN Lewis Structure. The number of steps can be adjusted depending on the molecule or ion’s complexity. To draw the Lewis structure of a molecule, there are a few general steps to follow. Steps to Remember While Drawing the Lewis Structure The hydrogen and nitrogen atoms surround the carbon atom, which is located in the middle. In addition, a triple bond is formed between the carbon and nitrogen atoms with a lone pair on nitrogen. HCN Lewis Structure contains a single bond between hydrogen and a carbon atom. The Octet Rule states that every atom likes to have eight valence electrons because this is the most stable and low-energy state. Valance electrons are electrons in the outermost shell that are vital for bonding and forming molecules. These valence electrons either share or are transferred (completely or partially) for bond formation. Only valence shell electrons involve in bond formation. Valence electrons are the total number of electrons present in the atom’s outermost shell. This blog article will help you understand the HCN lewis structure, hybridization, molecular geometry, and molecular orbital diagram.īefore going towards the HCN Lewis structure, let us understand some general terms used in it. It has a linear geometry with a triple bond between carbon (C) and nitrogen (N). The components of this weak acid are carbon, hydrogen, and nitrogen. It’s a liquid that has no color, is exceedingly combustible, and is poisonous. Prussic acid is another name for hydrogen cyanide or HCN. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
